Volume 10 | Issue 9 | September 2017

Release of VEGF from Dental Implant Improves Osteogenetic Process: Preliminary In Vitro Tests
Abstract
1. Introduction
2. Results
2.1. In Vitro Release of VEGF
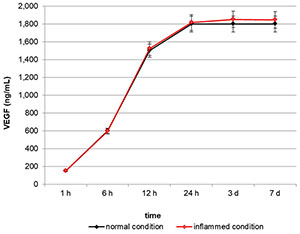
In order to test the ability of VEGF-enriched dental implants to release VEGF in vitro, we quantified the growth factor in the cell culture medium up to 7 days by means of an ELISA test. As reported in Figure 1, VEGF concentration increases in the medium in a time-dependent manner, starting from a basal concentration of 180 ng/mL after 1 h and reaching the maximum level of 1800 ng/mL after 24 h.
Figure 1. Quantification of Vascular Endothelial Growth Factor (VEGF) release in the cell culture medium. VEGF concentration increases in the medium in a time-dependent manner, reaching a plateau after 24 h.
2.2. Biocompatibility and Cell Proliferation
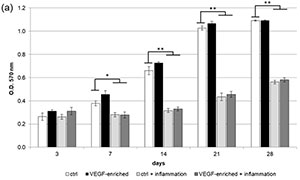
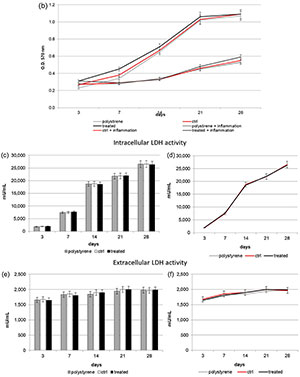
Figure 2. (a,b) 3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide (MTT) assay. (a: in form of bars and b: in form of line). Cells are able to proliferate on both the control and VEGF-enriched surface from 3 to 28 days of culture with no statistical difference between the two surfaces. On the contrary, when cells are treated with Tumor Necrosis Factor α (TNFα) their proliferation is significantly reduced on both the control and the VEGF-enriched implant; (c,d) Quantification of intracellular Lactate Dehydrogenase (LDH) activity (c: in form of bars; d: in form of lines). Intracellular LDH activity proves that cells are able to produce metabolites if seeded onto both surfaces, with improved results after 7 days from seeding; (e,f) Quantification of extracellular LDH activity (e: in form of bars; f: in form of lines). Extracellular LDH activity confirms that metabolites are secreted by the cells and are not associated with damage of the membrane. Statistically significant differences are indicated as * p < 0.05, ** p < 0.01, and compared with the control condition.
2.3. Intracellular and Extracellular Lactate Dehydrogenase (LDH) Activity
2.4. Reactive Oxygen Species (ROS) Production
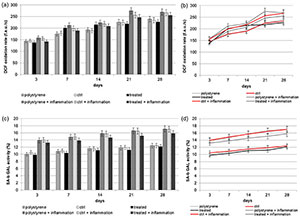
Figure 3. (a,b) (a: in form of bars and b: in form of line). Reactive Oxygen Species (ROS) production of Dental Pulp Stem Cells (DPSCs) seeded onto VEGF-enriched or control dental implants. Histograms show a slight time-dependent increase in metabolic activity in cells seeded onto both surfaces. The presence of VEGF on the implant surface is able to reduce the intracellular ROS production, especially under inflammatory conditions. Results are expressed as fluorescent arbitrary units per second (f.a.u./s); (c,d) (c: in form of bars and d: in form of line). Evaluation of Senescence-associated beta-galactosidase (SA-b GAL) (SA-b GAL) activity of DPSCs under normal or inflammatory conditions on surfaces combined or not with VEGF. Results show that cells cultured on implants loaded with VEGF have a lower SA-b GAL activity value compared to the cells seeded on the control one.
2.5. Senescence
2.6. Cell Morphology
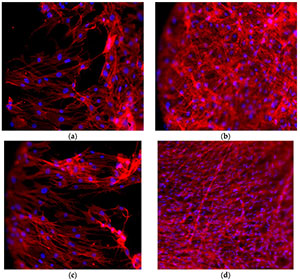
Figure 4. Immunofluorescence staining of the actin filament with phalloidin (in red). Cell nuclei are counterstained with Hoechst (in blue). (a for control; b on VEGF implants) After 7 days of culture, DPSCs have colonized the implant surface showing a star-like morphology; (c for control; d on VEGF implants) Cells are completely spread onto the implant surface after 14 days from seeding.
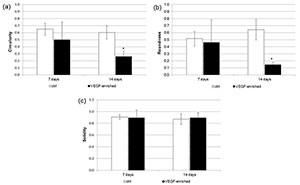
A detailed analysis of cell shape has additionally been performed. In particular, the Circularity, Roundness, and Solidity parameters have been considered. As shown in Figure 5, when cells were seeded onto the VEGF-enriched implants and cultured for 14 days, they assumed an elongated morphology, as suggested by the Circularity and Roundness calculated values. This would confirm that the cells lost the staminal rounded aspect for acquiring a more elongated morphology.
Figure 5. Analysis of the (a) Circularity; (b) Roundness; and (c) Solidity cell shape parameters after 7 and 14 days of culture onto the control and VEGF-enriched implants. * p < 0.05.
2.7. Effect of VEGF on Endothelial Cell Differentiation
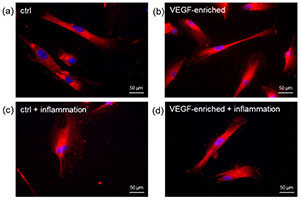
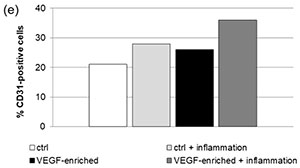
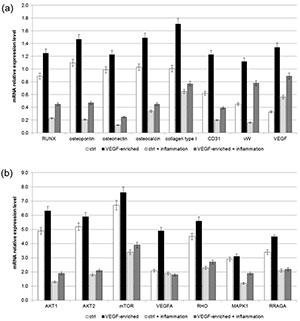
Figure 6. Immunofluorescence staining of the CD31 endothelial marker in DPSCs seeded onto (a) control implant; (b) implant loaded with VEGF; (c) control implant under inflammatory conditions; (d) VEGF-enriched implant under inflammatory conditions; (e) Evaluation of the % of CD31-positive cells.
2.8. Osteogenic Commitment
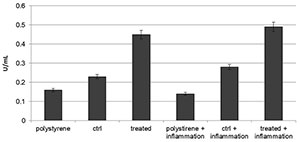
Figure 7. Alkaline phospahatase (ALP) production profiles in DPSCs seeded onto polystyrene, control or VEGF-enriched implants under normal or inflammatory conditions.
3. Discussion
4. Materials and Methods
4.1. Dental Implants
Dental implants were supplied by Ditron Dental (Ashkelon, Israel). In particular, MPI—Molecular Precision Implants with dimensions of 8 mm × 4.2 mm enriched with VEGF were used for the in vitro tests (Figure 9). The same implants without VEGF were used as control implants.
4.2. Quantitative Analysis of VEGF Release
4.3. DPSCs Isolation and Seeding onto Dental Implant
4.4. MTT Assay
4.5. LDH Activity
4.6. ROS Measurements
4.7. SA-b GAL Staining
4.8. Cell Shape Analysis
Cell shape was evaluated on samples stained for 40 min with 5 mg/mL phalloidin. Briefly, cells were fixed in 4% paraformaldehyde in PBS for 10 min, then permeabilized with 0.1% triton X-100 (Sigma-Aldrich, Saint Louis, MA, USA) in PBS for 30 min at room temperature. Phalloidin was then used for fluorescent staining of actin filaments, whilst nuclear staining was performed with 2 μg/mL Hoechst H33342 (Sigma-Aldrich) solution for 5 min. Images were acquired with the inverted optical microscope DMI4000 B (Leica Microsystems, Wetzlar, Germany). Then, ImageJ software (http://rsb.info.nih.gov/ij) was used to calculate cell area and different shape descriptors of at least 30 distinct cells. In detail, the Circularity (C), Roundness (R), and Solidity (S) of cells were calculated according to the following equations [42]:
(1)
Circularity = C = 4πA/P2
where A is the cell area and P is the perimeter;
(2)
Roundness = R = a/b
where a and b are the width and length of the minimum bounding, respectively;
(3)
Solidity = S = A/ConvexA
where ConvexA is the area enclosed by the smallest shell that borders all the points of the cell.
4.9. Immunofluorescence
4.10. Real-Time RT-PCR
4.11. Statistical Analyses
4.12. ALP Activity Measurements
Thereafter, 50 μL of 5 mM pNPP solution was added to each well containing test samples and background control and incubated for 60 min at 25 °C, protecting the plate from the light. A standard curve of 0, 4, 6, 12, 16 and 20 nmol/well was generated from 1 mM pNPP standard solution bringing the final volume to 120 μL with Assay Buffer. All reactions were then stopped by adding 20 μL of Stop solution into each standard and sample reaction except the sample background control reaction. Optical density was read at 405 nm in a microplate reader (Victor). The results were normalized subtracting the value derived from the zero standards from all standards, samples and sample background control. The pNP standard curve was plotted to identify the pNP concentration in each sample. ALP activity of the test samples was calculated as follow:
where A is the amount of pNP generated by samples (in μmol). V is the amount of sample added in the assay well (in mL). T is the reaction times (in minutes).
5. Conclusions
References
- Buser, D.; Sennerby, L.; De Bruyn, H. Modern implant dentistry based on osseointegration: 50 years of progress, current trends and open questions. Periodontol. 2000 2017, 73, 7–21. [Google Scholar] [CrossRef] [PubMed]
- Meng, H.W.; Chien, E.Y.; Chien, H.H. Dental implant bioactive surface modifications and their effects on osseointegration: A review. Biomark. Res. 2016, 4, 24. [Google Scholar] [CrossRef] [PubMed]
- Smeets, R.; Stadlinger, B.; Schwarz, F.; Beck-Broichsitter, B.; Jung, O.; Precht, C.; Kloss, F.; Gröbe, A.; Heiland, M.; Ebker, T. Impact of Dental Implant Surface Modifications on Osseointegration. Biomed. Res. Int. 2016, 2016, 6285620. [Google Scholar] [CrossRef] [PubMed]
- Reyes, C.D.; Petrie, T.A.; Burns, K.L.; Schwartz, Z.; García, A.J. Biomolecular surface coating to enhance orthopaedic tissue healing and integration. Biomaterials 2007, 28, 3228–3235. [Google Scholar] [CrossRef] [PubMed]
- Gongadze, E.; Kabaso, D.; Bauer, S.; Slivnik, T.; Schmuki, P.; Van Rienen, U.; Iglič, A. Adhesion of osteoblasts to a nanorough titanium implant surface. Int. J. Nanomed. 2011, 6, 1801–1816. [Google Scholar]
- Fraioli, R.; Rechenmacher, F.; Neubauer, S.; Manero, J.M.; Gil, J.; Kessler, H.; Mas-Moruno, C. Mimicking bone extracellular matrix: Integrin-binding peptidomimetics enhance osteoblast-like cells adhesion, proliferation, and differentiation on titanium. Colloids Surf. B Biointerfaces 2015, 128, 191–200. [Google Scholar] [CrossRef] [PubMed]
- Silva-Correia, J.; Zavan, B.; Vindigni, V.; Silva, T.H.; Oliveira, J.M.; Abatangelo, G.; Reis, R.L. Biocompatibility evaluation of ionic- and photo-crosslinked methacrylated gellan gum hydrogels: In vitro and in vivo study. Adv. Healthc. Mater. 2013, 4, 568–575. [Google Scholar] [CrossRef] [PubMed]
- Magdolen, U.; Auernheimer, J.; Dahmen, C.; Schauwecker, J.; Gollwitzer, H.; Tübel, J.; Gradinger, R.; Kesler, H.; Schmitt, M.; Diehl, P. Growth promoting in vitro effect of synthetic cyclic RGD-peptides on human osteoblast-like cells attached to cancellous bone. Int. J. Mol. Med. 2006, 17, 1017–1021. [Google Scholar] [PubMed]
- Figallo, E.; Flaibani, M.; Zavan, B.; Abatangelo, G.; Elvassore, N. Micropatterned biopolymer 3D scaffold for static and dynamic culture of human fibroblasts. Biotechnol. Prog. 2007, 23, 210–216. [Google Scholar] [CrossRef] [PubMed]
- Schliephake, H. Clinical efficacy of growth factors to enhance tissue repair in oral and maxillofacial reconstruction: A systematic review. Clin. Implant Dent. Relat. Res. 2015, 17, 247–273. [Google Scholar] [CrossRef] [PubMed]
- Ferroni, L.; Gardin, C.; Tocco, I.; Epis, R.; Casadei, A.; Vindigni, V.; Mucci, G.; Zavan, B. Potential for neural differentiation of mesenchymal stem cells. Adv. Biochem. Eng. Biotechnol. 2013, 129, 89–115. [Google Scholar] [PubMed]
- Verrecchia, F.; Mauviel, A. Transforming growth factor-beta signaling through the Smad pathway: Role in extracellular matrix gene expression and regulation. J. Investig. Dermatol. 2002, 118, 211–215. [Google Scholar] [CrossRef] [PubMed]
- He, H.; Yu, J.; Liu, Y.; Lu, S.; Liu, H.; Shi, J.; Jin, Y. Effects of FGF2 and TGFbeta1 on the differentiation of human dental pulp stem cells in vitro. Cell Biol. Int. 2008, 32, 827–834. [Google Scholar] [CrossRef] [PubMed]
- Farges, J.C.; Romeas, A.; Melin, M.; Pin, J.J.; Lebecque, S.; Lucchini, M.; Bleicher, F.; Magloire, H.J. TGF-beta1 induces accumulation of dendritic cells in the odontoblast layer. J. Dent. Res. 2003, 82, 652–656. [Google Scholar] [CrossRef] [PubMed]
- Scheufler, C.; Sebald, W.; Hülsmeyer, M. Crystal structure of human bone morphogenetic protein-2 at 2.7 A resolution. J. Mol. Biol. 1999, 287, 103–115. [Google Scholar] [CrossRef] [PubMed]
- Açil, Y.; Springer, I.N.; Broek, V.; Terheyden, V.; Jepsen, S. Effects of bone morphogenetic protein-7 stimulation on osteoblasts cultured on different biomaterials. J. Cell. Biochem. 2002, 86, 90–98. [Google Scholar] [CrossRef] [PubMed]
- Saito, T.; Ogawa, M.; Hata, Y.; Bessho, K.J. Acceleration effect of human recombinant bone morphogenetic protein-2 on differentiation of human pulp cells into odontoblasts. J. Endod. 2004, 30, 205–208. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Gluhak-Heinrich, J.; Martinez, M.; Li, T.; Wu, Y.; Chuang, H.H.; Chen, L.; Dong, J.; Gay, I.; MacDougall, M. Bone morphogenetic protein 2 mediates dentin sialophosphoprotein expression and odontoblast differentiation via NF-Y signaling. J. Biol. Chem. 2008, 283, 19359–19370. [Google Scholar] [CrossRef] [PubMed]
- Solchaga, L.A.; Penick, K.; Porter, J.D.; Goldberg, V.M.; Caplan, A.I.; Welter, J.F. FGF-2 enhances the mitotic and chondrogenic potentials of human adult bone marrow-derived mesenchymal stem cells. J. Cell. Physiol. 2005, 203, 398–409. [Google Scholar] [CrossRef] [PubMed]
- Shimabukuro, Y.; Ueda, M.; Ozasa, M.; Anzai, J.; Takedachi, M.; Yanagita, M.; Ito, M.; Hashikawa, T.; Yamada, S.; Murakami, S. Fibroblast growth factor-2 regulates the cell function of human dental pulp cells. J. Endod. 2009, 35, 1529–1535. [Google Scholar] [CrossRef] [PubMed]
- Joseph, B.K.; Savage, N.W.; Young, W.G.; Gupta, G.S.; Breier, B.H.; Waters, M.J. Expression and regulation of insulin-like growth factor-I in the rat incisor. Growth Factors 1993, 8, 267–275. [Google Scholar] [CrossRef] [PubMed]
- Feng, X.; Huang, D.; Lu, X.; Feng, G.; Xing, J.; Lu, J.; Xu, K.; Xia, W.; Meng, Y.; Tao, T.; et al. Insulin-like growth factor 1 can promote proliferation and osteogenic differentiation of human dental pulp stem cells via mTOR pathway. Dev. Growth Differ. 2014, 56, 615–624. [Google Scholar] [CrossRef] [PubMed]
- Ferrara, N. Vascular endothelial growth factor: Basic science and clinical progress. Endocr. Rev. 2004, 25, 581–611. [Google Scholar] [CrossRef] [PubMed]
- D’ Alimonte, I.; Nargi, E.; Mastrangelo, F.; Falco, G.; Lanuti, P.; Marchisio, M.; Miscia, S.; Robuffo, I.; Capogreco, M.; Buccella, S.; et al. Vascular endothelial growth factor enhances in vitro proliferation and osteogenic differentiation of human dental pulp stem cells. J. Biol. Regul. Homeost. Agents 2011, 25, 57–69. [Google Scholar]
- D’Aquino, R.; Graziano, A.; Sampaolesi, M.; Laino, G.; Pirozzi, G.; De Rosa, A.; Papaccio, G. Human postnatal dental pulp cells co-differentiate into osteoblasts and endotheliocytes: A pivotal synergy leading to adult bone tissue formation. Cell Death Differ. 2007, 14, 1162–1171. [Google Scholar] [CrossRef] [PubMed]
- Gardin, C.; Bressan, E.; Ferroni, L.; Nalesso, E.; Vindigni, V.; Stellini, E.; Pinton, P.; Sivolella, S.; Zavan, B. In vitro concurrent endothelial and osteogenic commitment of adipose-derived stem cells and their genomical analyses through comparative genomic hybridization array: Novel strategies to increase the successful engraftment of tissue-engineered bone grafts. Stem Cells Dev. 2012, 21, 767–777. [Google Scholar] [CrossRef] [PubMed]
- Duan, X.; Murata, Y.; Liu, Y.; Nicolae, C.; Olsen, B.R.; Berendsen, A.D. Vegfa regulates perichondrial vascularity and osteoblast differentiation in bone development. Development 2015, 142, 1984–1991. [Google Scholar] [CrossRef] [PubMed]
- Hu, K.; Olsen, B.R. Osteoblast-derived VEGF regulates osteoblast differentiation and bone formation during bone repair. J. Clin. Investig. 2016, 126, 509–526. [Google Scholar] [CrossRef] [PubMed]
- Berendsen, A.D.; Olsen, B.R. How vascular endothelial growth factor-A (VEGF) regulates differentiation of mesenchymal stem cells. J. Histochem. Cytochem. 2014, 62, 103–108. [Google Scholar] [CrossRef] [PubMed]
- Albrektsson, T.; Wennerberg, A. Oral implant surfaces: Part I. review focusing on topographic and chemical properties of different surfaces and in vivo responses to them. Int. J. Prosthodont. 2004, 17, 536–543. [Google Scholar] [PubMed]
- Novaes, A.; Papalexiou, V.; Grisi, M.; Souza, S.; Taba, M.; Kajiwara, J. Influence of implant microstructure on the osseointegration of immediate implants placed in periodontally infected sites. A histomorphometric strudy in dogs. Clin. Oral Implants Res. 2004, 15, 34–43. [Google Scholar] [CrossRef] [PubMed]
- Burgos, P.; Rasmusson, L.; Meirelles, L.; Senerby, L. Early bone tissue response to turned and oxidized implants in a rabbit tibia. Clin. Implant Dent. Relat. Res. 2008, 10, 181–190. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Xiropaidis, A.; Sorensen, R.; Albandar, J.; Hall, J.; Wikesjo, U. Bone formation of titanium porous oxide (TiUnite) oral implants in type IV bone. Clin. Oral Implants Res. 2005, 16, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Mendes, V.; Moineddin, R.; Davies, J. The effect of discrete calcium phosphate nanocrystals on bone-bonding to titanium surfaces. Biomaterials 2007, 28, 4748–4755. [Google Scholar] [CrossRef] [PubMed]
- Mendes, V.; Moineddin, R.; Davies, J. Discrete calcium phosphate nanocrystalline deposition enhances osteoconduction on titanium based implant surfaces. J. Biomed. Mater. Res. A 2009, 90, 577–585. [Google Scholar] [CrossRef] [PubMed]
- Kempen, D.H.; Lu, L.; Heijink, A.; Hefferan, T.E.; Creemers, L.B.; Maran, A.; Yaszemski, M.J.; Dhert, W.J. Effect of local sequential VEGF and BMP-2 delivery on ectopic and orthotopic bone regeneration. Biomaterials 2009, 30, 2816–2825. [Google Scholar] [CrossRef] [PubMed]
- Kaigler, D.; Wang, Z.; Horger, K.; Mooney, D.J.; Krebsbach, P.H. VEGF scaffolds enhance angiogenesis and bone regeneration in irradiated osseous defects. J. Bone Miner. Res. 2006, 21, 735–744. [Google Scholar] [CrossRef] [PubMed]
- Leach, J.K.; Kaigler, D.; Wang, Z.; Krebsbach, P.H.; Mooney, D.J. Coating of VEGF-releasing scaffolds with bioactive glass for angiogenesis and bone regeneration. Biomaterials 2006, 27, 3249–3255. [Google Scholar] [PubMed]
- Bressan, E.; Ferroni, L.; Gardin, C.; Pinton, P.; Stellini, E.; Botticelli, D.; Sivolella, S.; Zavan, B. Donor age-related biological properties of human dental pulp stem cells change in nanostructured scaffolds. PLoS ONE 2012, 7, e49146. [Google Scholar] [CrossRef] [PubMed]
- Zulato, E.; Favaretto, F.; Veronese, C.; Campanaro, S.; Marshall, J.D.; Romano, S.; Cabrelle, A.; Collin, G.B.; Zavan, B.; Belloni, A.S.; et al. ALMS1-deficient fibroblasts over-express extra-cellular matrix components, display cell cycle delay and are resistant to apoptosis. PLoS ONE 2011, 6, e19081. [Google Scholar] [CrossRef] [PubMed]
- Zavan, B.; Giorgi, C.; Bagnara, G.P.; Vindigni, V.; Abatangelo, G.; Cortivo, R. Osteogenic and chondrogenic differentiation: Comparison of human and rat bone marrow mesenchymal stem cells cultured into polymeric scaffolds. Eur. J. Histochem. 2007, 51, 1–8. [Google Scholar] [PubMed]
- Rocca, A.; Marino, A.; Rocca, V.; Moscato, S.; de Vito, G.; Piazza, V.; Mazzolai, B.; Mattoli, V.; Ngo-Anh, T.J.; Ciofani, G. Barium titanate nanoparticles and hypergravity stimulation improve differentiation of mesenchymal stem cells into osteoblasts. Int. J. Nanomed. 2015, 10, 433–445. [Google Scholar]
- Pfaffl, M.W. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res. 2001, 29, e45. [Google Scholar] [CrossRef] [PubMed]